Quantaflex ELTM Design Guide
Photometric Characteristics
Available Colors - What determines the Color of an EL Lamp?
What determines the Color of an EL Lamp?
The type of Phosphor used.
Each phosphor particle is a semi-conductor comprised of a “doped” Zinc Sulfide crystal.
The type and concentration of dopant in the crystal determined the wavelength of the emitted photons.
There are numerous different phosphors utilized to manufacture Quantaflex TM
lamps, each one having its own unique spectral output.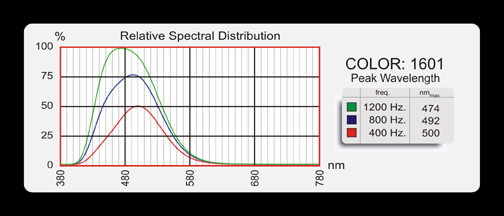
The frequency of the AC signal applied.
As the frequency of the applied power is increased, the frequency of the light emitted is also slightly increased (blue shift).
The extent to which the spectral output is effected by the input is dependent upon the type of phosphor used. Some phosphors are dramatically effected, others only minimally. See Photometric Data for data on specific colors.

The addition of Fluorescent
Conversion Dyes and Pigments to the Phosphor Layer or the addition
of Conversion Dyes or Filters to the front of the lamp.
This method is used to produce “Dye Conversion White” colors. There
are no commercially available phosphors which efficiently produce
light in the red-ora nge-yellow spectrum.
Therefore, fluorescent conversion dyes are often used to produce the
red and yellow components necessary to produce various shades of
white light. These red and yellow fluorescent dyes and pigments can
absorb some of the green and blue light produced by phosphors which
are efficient in this range and re-emit the light in the Red-Yellow
spectrum. The choice of dye and its concentration will determine the
shade of white produced. Because some of the energy is lost in this
color conversion, the efficiency of dye conversion colors is
generally less than those in the Aviation Green/Aviation Blue
spectrum.
nge-yellow spectrum.
Therefore, fluorescent conversion dyes are often used to produce the
red and yellow components necessary to produce various shades of
white light. These red and yellow fluorescent dyes and pigments can
absorb some of the green and blue light produced by phosphors which
are efficient in this range and re-emit the light in the Red-Yellow
spectrum. The choice of dye and its concentration will determine the
shade of white produced. Because some of the energy is lost in this
color conversion, the efficiency of dye conversion colors is
generally less than those in the Aviation Green/Aviation Blue
spectrum.
Back | Top of Page | Life Characteristics